Chirality And Resolution
Chirality can be defined as the potential of a molecule to occur in two asymmetric forms that are non-superimposable mirror images of each other without changing the atomic composition, atom-atom connections, or bond orders for example all natural amino acids are in L-form and its mirror image is unnatural D-form. These two versions of the molecule are referred to as enantiomers. Similarly, all natural sugars are D-form (e.g. D-glucose) and their counter enantiomers are unnatural L-sugars. Because our body building blocks are chiral it perceive chiral chemicals differently based on their enantiomeric form. Hence the taste, smell and drug activities may differ significantly depending upon their enantiomeric form (please see the Table).
Because of these inherent properties of chiral compounds it has become mandatory to synthesise these molecules in their desired enantiomeric form in high optical purity efficiently and economically for commercialization.
Therefore in the last 4 decades asymmetric synthesis, asymmetric catalysis and optical resolution have become essential tools in relevant Chemical Industries.
Chirality vis-a-vis activity (Table)
| S.No | Drug/ Chemical | S Form | R Form |
|---|---|---|---|
| 1 | Chloramphenicol | Inactive | Antibacterial |
| 2 | Ethambutol | Tuberculostatic | Causes Blindness |
| 3 | Carvone | Caraway flavour | Spearmint flavour |
| 4 | Methyl epijasmate | Less fragrant | 4 times stronger |
| 5 | Asparagine | Bitter taste | Sweet taste |
| 6 | Propranolol | b-blocking agent | 100 Times less |
| 7 | Fluazifop | Inactive | Herbicide |
| 8 | Paclobutraz | Plant growth regulator | Fungicide |
| 9 | Warfarin | Hypoprothrombinaemic agent | 5-6 Times less active |
| 10 | Clozylacon | Fungicide (2S,3R form) | Inactive (2R,3S) |
Kinetic Resolution
Kinetic resolution is one of the most powerful method for achieving chiral compounds in high optical purity from the racemic mixture. This method is based on the fact that individual enantiomers are transformed to a product at different rates or in some cases only one enantiomer reacts and at the end of the reaction the other enantiomer is recovered unchanged. Many procedures based on catalytic kinetic resolution have been developed for alcohols, epoxides, amines, carbonyl and sulfur containing compounds.
Among these Hydrolytic Kinetic Resolution (HKR) and Aminolytic Kinetic Resolution (AKR) of racemic epoxides and Oxidative Kinetic Resolution (OKR) of racemic alcohols have been successfully applied at commercial scale with the intervention of some very powerful and rugged chiral catalysts.
Chiral Salen complexes with different metal ions have shown remarkable catalytic activity in various asymmetric organic transformations.
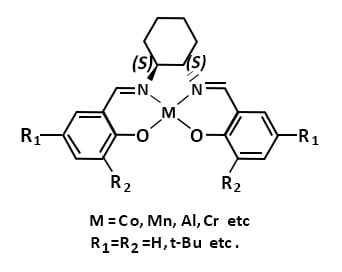
For example, Chiral SALEN ligands derived from 1,2-diaminocyclohexane are used as catalyst in variety of asymmetric organic transformations as represented below.
Cobalt Complexes: in Hydrolytic Kinetic Resolution (HKR) of racemic terminal epoxides, asymmetric polymerization, alkylation and oxidation.
Manganese complexes: in Asymmetric epoxidation of non-functionalized olefins, oxidative kinetic resolution (OKR) of racemic secondary alcohols and sulfoxidation.
Aluminum complexes: Asymmetric alkylation of aldehydes and ketones, asymmetric polymerization of epoxides.
Chromium complexes: Asymmetric epoxidation
Some of the examples of drugs derived from chirally pure epichlorohydrin, glycidol and 3-chloro-1,2-propanediol are given below.
From (S)-epichlorohydrin
Landiolol, Rivaroxaban, Velusetrag, Amitifadine, Sutezolid, Linezolid, (S)-oxiracetam, S-Propranolol
From (S)-glycidol
Levobunolol, Tenofovir, Indinavir sulfate
From (R)-3-chloropropane-1,2-diol
Pretomanid, L-carnitine, Levodropropizine
From (R)-epichlorohydrin
Levobetaxolol, Levomilnacipran, Lemborexant, Inakalant, (R)-1,4-dioxan-2yl-methanamine
From (R)-glycidol
(S)-(-)-Atenolol, (S)-(-)-metroprolol, Levodropropizine